Event - QualityJam - 2018.10.18 - London, UK
Date: 18 October 2018. Time:09:00-17:00.
Place: The Brewery, 52 Chiswell St, London EC1Y 4SD, UK
Duration: 8 hours.
Language: English.
Audience: 100-150 people .
Ways to Attend:
| 1. Personally - visit the meeting: | The Brewery, 52 Chiswell St, London EC1Y 4SD, UK |
| 2. Skype - join remote: | not available |
|

|
Websites: https://www.qualityjam.com/uk/
GARANTIS IT Solutions Ltd. takes part in this event to share information and experience about Polarion projects in Testing/QA area.
This Conference is a good chance for QA Managers, Test Engineers, Testing Companies to share experience, find solutions, learn new ways to solve common tasks in the area of QA/Testing.

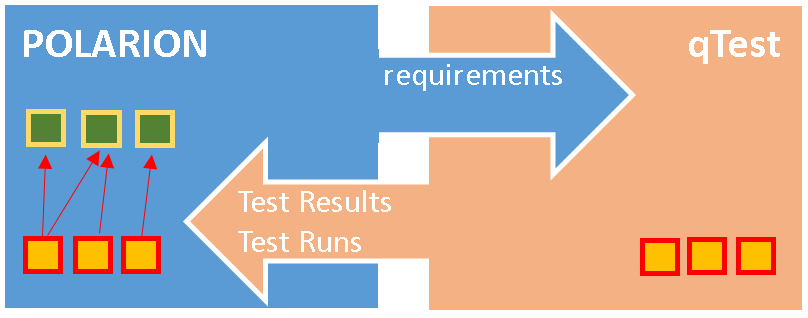
ANNOUNCED new integration qTest + Polarion ALM
We are proud to announce our new product - integration to support test execution in qTest without loosing traceability and coverage in Polarion ALM.
Features:
- copy Test Cases to qTest
- copy results back to Polarion
- preserve links in Polarion
- execute in qTest
- view reports in Polarion and in qTest
Go to Product page...
|
|
|
REVIEW of the Conference
SUMMARY:
- Discussions of Testing and QA topics. General information about Testing.
- Testing in Agile projects - exchange of experience.
- Personal development of Test Engineers. Many come NOT from Testing background. How to educate them?
- Networking, networking, networking... Meeting people and talking. Attendees were from United Kingdom, Germany, Netherlands, Denmark, United States.
Networking, meeting people, exchanging ideas and experiences...

TALKS
- about TOOLs
- about EXPERIENCES (bad and good)
- about HISTORY of Projects
- about qTest and Tricentis, past and future
Success of QA processes and partners - how to assess?

Agile, Waterfall, Wagile..??

qTest and Tricentis, past and future

History of Testing - 1979 - first definition of BUG and TEST CASE

Meet us after the Conference
We are always available to support our clients and to give free advise:
E-mail: info@garantis-solutions.com
Direct Phone: +44 (0) 118 343 1584
AEROSPACE &
DEFENCE |
AUTOMOTIVE
SOLUTIONS |
MEDICAL
DEVICES |
ENTERPRISE
AGILE |
EMBEDDED
SYSTEMS |
 |
 |
 |
 |
 |
| Reduce time-to-market and improve quality and safety across multiple, complex systems. |
Collaborate across complex internal and supplier ecosystems. Accelerate Functional Safety compliance for ISO 26262, Automotive SPICE. and CMMI standards. |
Accelerate Medical Device Innovation and Automate Proof of Compliance. |
Achieve agility while protecting quality and compliance regulations. Keep your approval process as lean as possible but still compliant. |
Automate management of systems engineering projects and develop quality embedded software with the industry's highest efficiency and ROI. |